The relevance of extensive RNA editing in tumor evolution
Elevated RNA Editing Activity Is a Major Contributor to Transcriptomic Diversity in Tumors
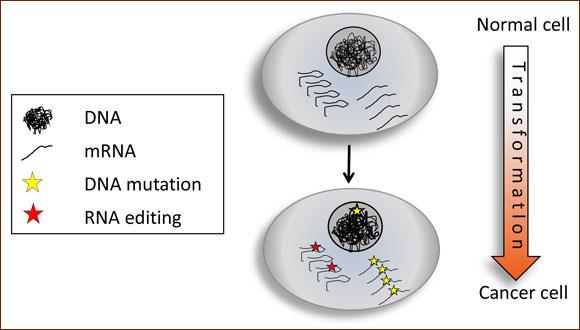
Cancer is driven by alterations of the genetic information, most commonly through mutations in DNA that deregulate normal cellular functions and push cell growth into overdrive. However, DNA mutations are not the only way to alter genetic sequence information. During the past decade, computational analysis of extensive sequencing data has revealed that RNA editing, a process through which changes are made to the nucleotide sequences of RNAs, is a widespread phenomenon, with more than a million sites of editing detected so far in humans. Now, three independent groups of investigators, one of them led by Erez Levanon (Bar-Ilan U.) and Eli Eisenberg (from the School of Physics & Astronomy at TAU), have revealed that RNA editing levels are significantly altered across cancer types, changing the RNA messages from their normal pattern, or, in other words, introducing RNA mutations that alter the genomic information in a tumor-specific way.
RNA edits are similar to DNA mutations in that most occurrences are simply "passengers", having no functional meaning and no role in tumor evolution. However, a small fraction of the mutations may be "drivers" - modulating RNA or protein function in a clinically significant way, promoting cancer progression. Indeed, manipulating editing levels in cells affects cell survival, proliferation and apoptosis, indicating that A-to-I editing may contribute to the uncontrolled growth observed in cancer. In some cancers higher levels of editing in patients corresponded with less favorable clinical outcomes.
These findings highlight the importance of a new class of mutations, RNA mutations, in understanding cancer biology, and open new directions for diagnostics and prognostic purposes. In addition, identifying the proteins or pathways which are consistently mutated at the RNA level could offer new targets for therapeutics, including the possibility of guiding the endogenous RNA editing mechanism to 'correct' these mutations.
Further details and the full publication in ScienceDirect.